Description and Composition of Mariprist
Mariprist is a combination drug which contains Mifepristone and Misoprostol as its active ingredients. Clearly put, Mariprist is not formulated into a fixed dose combination. It is only packaged together as they are often used together for Pregnancy termination. More details on usage is written below.
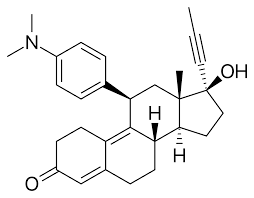
Mifepristone is a synthetic steroid with an anti-progestational action as a result of competition with progesterone at the progesterone receptors. It is almost exclusively used for termination of pregnancy.
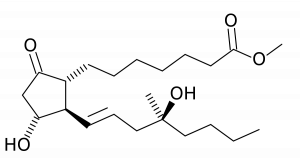
Misoprostol is a synthetic prostaglandin E1 analogue. In comparison to other prostaglandin analogues, misoprostol is stable at room temperature, and has fewer side effects. Its clinical applications include medical abortion, treatment of incomplete abortion and miscarriages, cervical priming prior to surgical procedures, induction of labour, and prevention and treatment management of postpartum haemorrhage. It is also used in the prevention and treatment of peptic ulcer disease.
The plasma elimination half-life for mifepristone is 30-40 hours, and for misoprostol is 20-40 minutes. After oral administration, mifepristone is rapidly and almost completely absorbed. Plasma concentrations peak at about 45 minutes.
Mifepristone is primarily metabolised by CYP3A4 enzyme. Following sublingual administration, misoprostol dissolves and is absorbed rapidly, with peak concentrations occurring at about 30 minutes. Bioavailability is very high.
Indication of Mariprist
The combination is used for medical termination of intra-uterine pregnancy of up to 63 days (9 weeks) gestation.
Dosage of Mariprist
For medical termination of intra-uterine pregnancy of up to 63 days gestation:
Adult And Adolescent
- Mifepristone 200 mg orally as a single dose, followed 36-48 hours later by misoprostol 800 µg by vagina and individual observed for at least 6 hours (or until bleeding or pain at acceptable level) with follow-up visit 10-15 days later to verify complete expulsion (if treatment fails essential that pregnancy terminated by another method).
How to Use Mariprist
- Swallow one tablet of Mifepristone (200 mg) as a single dose with a glass of water.
- Wait for 24-48 hours
- Take 4 tablets of Misoprostol (800 μµg) as a single dose, sublingually or vaginally
For buccal dose:
- Place the 4 Misoprostol tablets (as a single 800 mcg buccal dose) between your cheeks and gums and allow to dissolve for 30 minutes.
If there are still some tablet pieces left in your mouth after 30 minutes, swallow them with a glass of water. Plan to rest for 3 hours after taking the Misoprostol tablets.
The medications may not work as well if you take misoprostol sooner than 24 hours after taking mifepristone or later than 48 hours after taking mifepristone.
Paracetamol or Ibuprofen may be taken to reduce pain. Applying a heat pack or hot water bottle to below the stomach may help to ease the pain. Patient are to seek immediate medical attention if they experience severe or persistent abdominal pain or cramping, heavy bleeding or no bleeding, high fever, excess vomiting or diarrhoea.
Contraindications of Mariprist
When not to use a combination of Mifepristone and Misoprostol to terminate pregnancy include the following:
- Avoid if pregnancy is more than 9 weeks.
- Uncontrolled severe asthma.
- Suspected ectopic pregnancy (use other specific means of termination).
- Chronic adrenal failure, porphyria.
- Intra-uterine contraceptive device (IUD)
Precautions
The following are precautions you are to observe While Taking Mifepristone and Misoprostol
- If treatment fails, terminate pregnancy using another method.
- Don’t use in a patient with history of caesarean section or major uterine surgery, grand multiparas (risk of rupture) or use with caution
- Asthma (avoid if severe).
- Use with caution in haemorrhagic disorders and anticoagulant therapy
- Use with caution in prosthetic heart valve or history of endocarditis (antibacterial prophylaxis recommended).
- Use with caution in smokers aged over 35 years (increased risk of cardiovascular events).
- Use with caution in adrenal suppression (may require corticosteroid).
- Not recommended in hepatic or renal impairment.
- Avoid aspirin and NSAIDs for analgesia
Pregnancy & Lactation
Recommendations on the use of Mifepristone + Misoprostol in Pregnancy & Lactation:
Mifepristone:
Mifepristone is used as abortifacient and so contraindicated in wanted pregnancy. However, no significant association with teratogenicity has been reported among pregnancies carried to term after failed termination attempts with mifepristone. There is paucity of data on safety of mifepristone during breastfeeding. No toxic effect has been reported so far among breastfed infants following accidental intake by the lactating mothers
Misoprostol:
Misoprostol has stimulant effect on the uterus causing miscarriage. It also has teratogenic effects and, therefore, contraindicated during pregnancy. Misoprostol can induce diarrhoea in the breastfeeding infant, otherwise no report of other significant adverse effects.
Interactions of Mariprist with Other Drugs and Foods
- Magnesium-containing antacids: May worsen diarrhoea associated with Misoprostol
- CYP3A4 Inhibitors & Inducers: Caution when used with these agents.
- Mifepristone is primarily metabolised by CYP3A4 enzyme. Examples include azole antifungals (itraconazole, ketoconazole); macrolide antibiotics (clarithromycin, erythromycin); rifamycins (rifampicin); AEDs (carbamazepine, phenytoin), etc.
Adverse effects of Mariprist
Nausea, vomiting, gastrointestinal cramps; uterine hyperstimulation, uterine rupture, vaginal bleeding (sometimes severe), fetal distress.
Less commonly hypersensitivity reactions including rash, urticaria, and facial oedema. Rarely malaise, headache, fever, hot flushes, dizziness, chills.
Storage of Mariprist
Store in a cool and dry place. Keep away from light and the reach of children.
Chemical Structures